How do the strawberries smell?
 |
For sure, beautifully! especially when we taste them in December! And how do they smell in summer? Differently? |
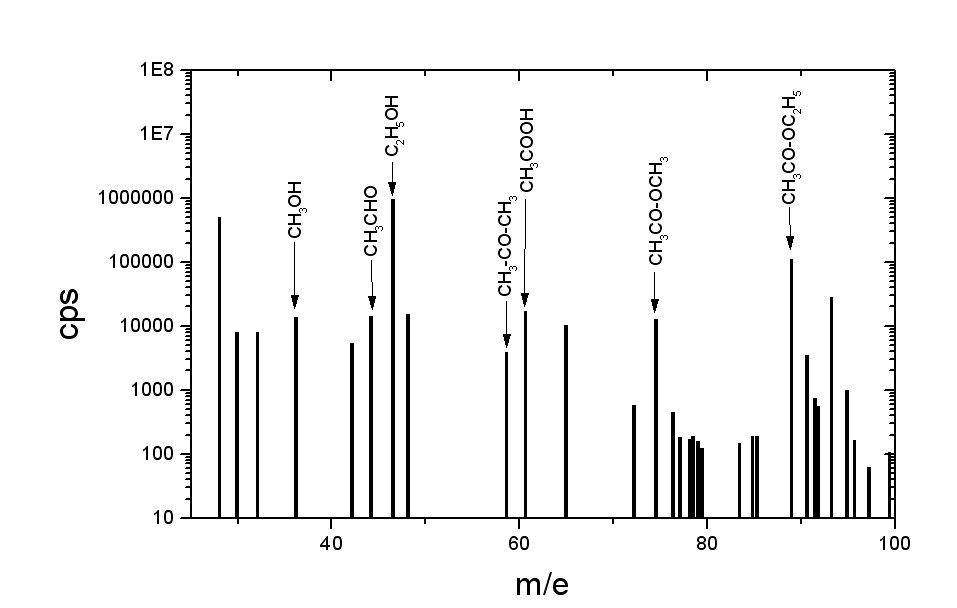
The flavour (i.e. its mass spectrum) of fresh strawberries, from proton transfer mass spectrometer.
Analysis of the spectra was performed only for the simplest chemical species like methanol, ethanol, acethyl-aldehyde, acetone, acetic acid, methyl acetate and ethyl acetate. Methyl and ethyl alcohols there can be treated as "vectors", carrying another smelling substances. Methyl and ethyl octanes have a pleasant flavour, rest of the substances are
odorous.
One of the main problems in mass spectrometry is how to ionize a molecule, without destroying it. Only ionized (i.e. those which lost or gained an electron) atoms and/or molecules and/or radicals can be trapped and transported by (relatively) weak electric and magnetic fields.
But ionization by collision with electrons or photoionization usually breaks the parent molecule in many fragments. The new, “soft” ionization, uses proton transfer of a proton from the ionized H3O+ molecule to the searched molecule. This transfer does not destroy the parent molecule and allows the flavour of strawberries to be measured.
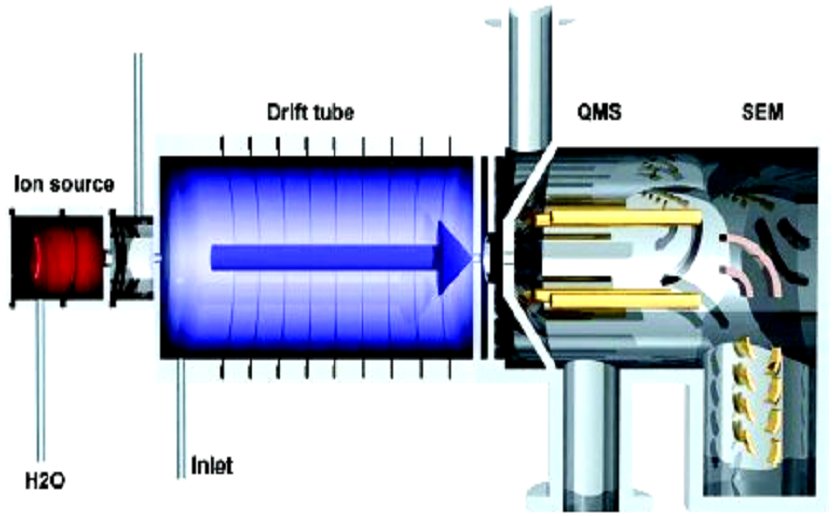 |
 |
Proton-transfer-reaction mass spectroscopy |
This was the transfer of proton, but also transfer by proton exists. |