Carbon balls
We all know that graphite and diamond are two carbon forms: one is grey, fragile, made of slipping hexagons, another brilliant, cubic and hard.
But what about a common sooth from a chimney? It is black and seems fatty.
Only in 1985, R.F. Cure, H. W. Kroto, and R. E. Smalley discovered in deposits from carbon electric arc a new form, an empty cage, made of 60 carbon atoms, called fullerene. Now it is a whole family.
Fullerenes were under study for potential medicinal use — binding specific antibiotics to the structure to target resistant bacteria and even target certain cancer cells such as melanoma.
Attaching potassium, lithium or rubidium atoms they become superconductors.
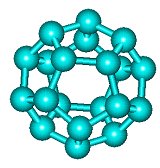
C20
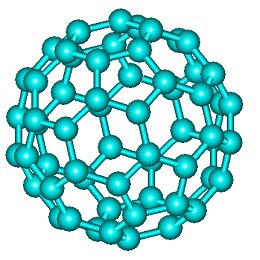
C60
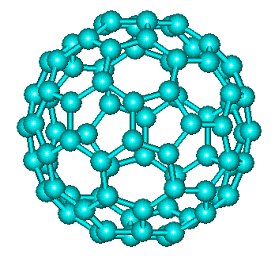
C80
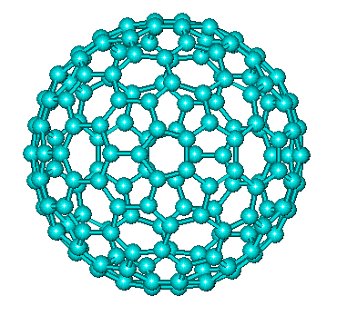
C180
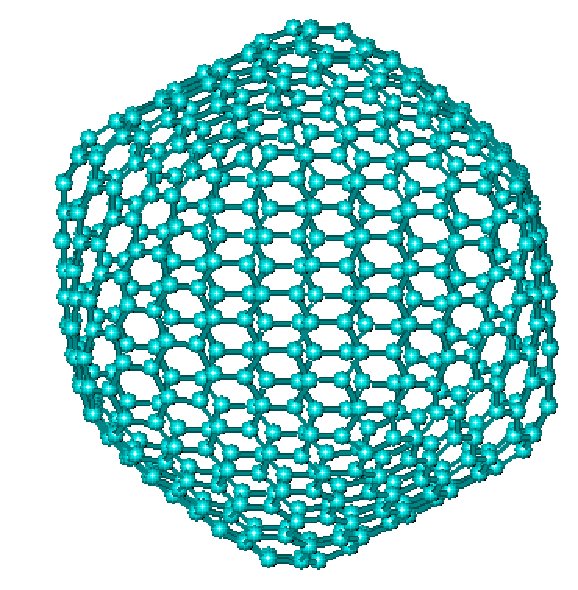
C540
The comparatively stable C60 was the first to be discovered, but several other members of the family have now been made. Big fullerenes are no more spherical.
next